Carbon Dioxide Physical State At Room Temperature
55 c 5 4 bar bar upper limit.
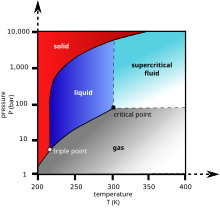
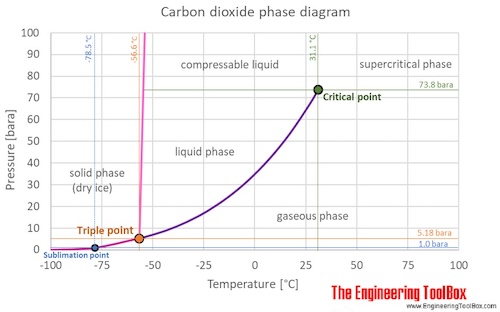
Carbon dioxide physical state at room temperature. At 1 atmosphere near mean sea level pressure the gas deposits directly to a solid at temperatures below 78 5 c 109 3 f. Lower limit for calculation. Answered september 15 2016 at room temperature carbon dioxide co2 is a colorless odorless faintly acidic tasting non flammable gas. Carbon dioxide has no liquid state at pressures below 5 1 standard atmospheres 520 kpa.
30 c 72 14 bar. It can only exist at a pressure above 5 1 atm 5 2 bar. Calculation of thermodynamic state variables of carbon dioxide at saturation state boiling curve. Carbon dioxide is a gas at standard conditions.
Liquid carbon dioxide is the liquid state of carbon dioxide which cannot occur under atmospheric pressure. The phase diagram for carbon dioxide shows the phase behavior with changes in temperature and pressure. The answer to your question is in the molecular structure of co2 there are 4 electrons in outer orbit of carbon and six in that of oxygen. 194 7 k and the solid sublimes directly to a gas above 78 5 c.
The following thermodynamic properties will be calculated.